Our group focusits research on new biomedical nanotechnology-based applicationsto develop new innovative drug delivery therapies and diagnostic systems to provide solutions to unmet medical needs in the oncology field and also in rare diseases. In particular, we use specific tailor-made imaging-based in vitro and in vivo models (bioluminescent and fluorescent models) to identify new therapeutic targets and to address the preclinical validation of therapeutic nanoconjugates and biosensors, in these areas.Briefly,
- Specific bioluminescent cancer stem cell models have been generated to develop new actively targeted nanotherapies able to sensitize cancer stem cells to current treatments, and to inhibit the reversion phenotype from non-cancer stem cells to cancer stem cells.
- Specific drug delivery systems are being tested as new enzyme replacement therapies for lysosomal storage diseases.
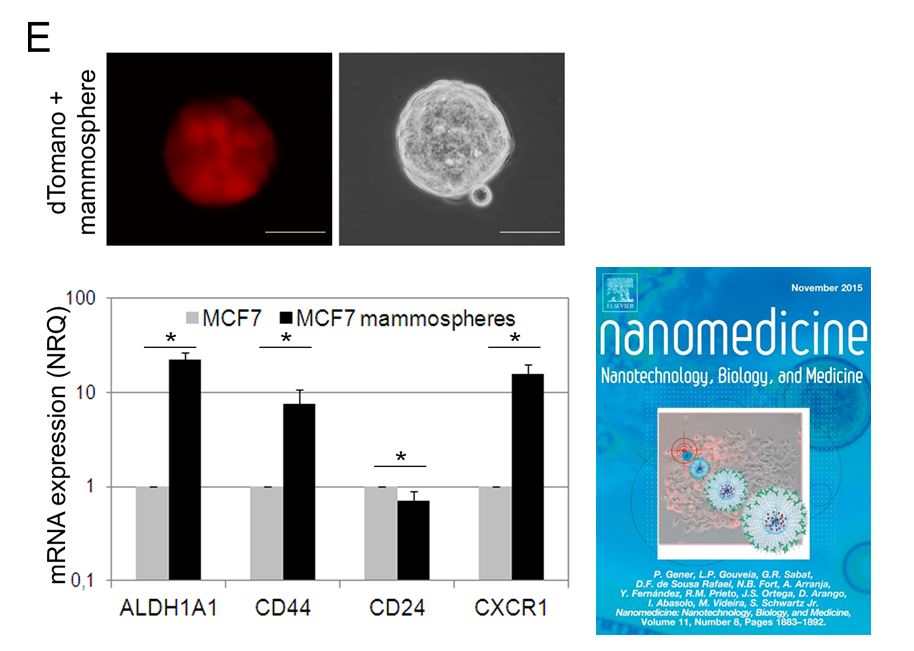
Fig1. New MCF7-ALDH1A1/tdTomatoCSCmodel. After transfectionwith the reporter vectorALDH1A1/tdTomato, expression of tdTomatowas detected only in the CSC subpopulation. Further, tdTomato+ CSC were also able to form mammospheres in low attachment plates and in serum free media. These mammospheres showed overexpression of the CSC markers ALDH1A1, CD44 and CXCR1 (22.2 ± 4.8; 7.5 ± 3.4; 15.5 ± 4.1 fold, respectively) and a lower expression of CD24 (0.7 ± 0.12 fold) compared to tdTomato− cells.(Nanomedicine: Nanotechnology, Biology, and Medicine 11 (2015) 1883–1892)
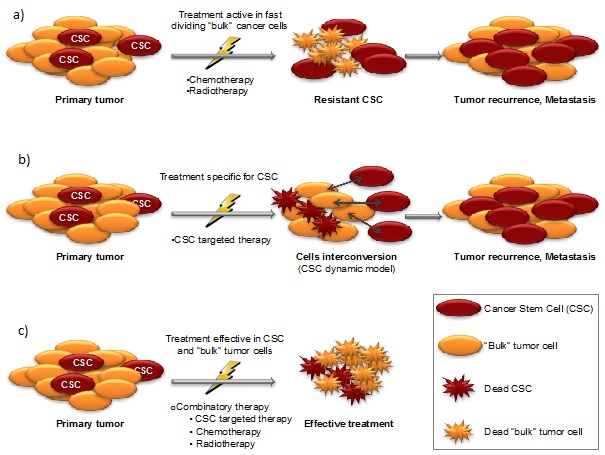
Fig 2. CSC can self-renew, have long-term propagating capacity, and can generate tumor. Nonetheless, CSC can also be de-differentiated (reversion phenotype) from “bulk” tumor cells, to guarantee tumor progression.Just therapies targeting CSC and “bulk” tumor cells as well as the reversion phenotype will effectively eradicate cancer (Nanomedicine (Lond.) (2016) 11(3), 307–320).
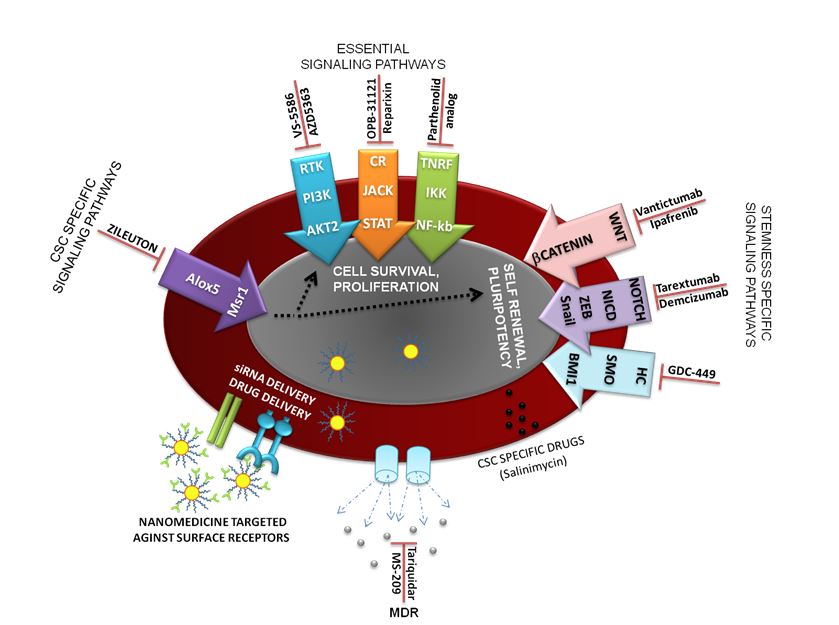
Fig.3. CSC therapies could be designed against i.) Essential cellular signaling pathways, ii.) Stemness specific pathways and iii.) CSC specific pathways. The delivery of therapeutic compounds could avoid multi drug resistance (MDR) channels by targeted delivery into the CSC(Nanomedicine (Lond.) (2016) 11(3), 307–320).
more info







