welcome
CIBBIM-NANOMEDICINE
Vall d’Hebron Institut de Recerca
CIBBIM-Nanomedicine Director
Dra. Ibane Abasolo Olaortúa
Nanomedicine and Advanced Therapies as translational sciences have the ultimate goal to provide cost effective novel therapies and diagnostics. As key enable technologies these new options will cause profound changes in healthcare, making feasible more personalized, predictive, preventive, and regenerative medicine.
Events and Conferences
PRECLINICAL SERVICES
This platform offers standard in vitro experimentation procedures for the functional validation of candidate genes and biomarkers, and for the efficacy and toxicological evaluation of new drugs, biomaterials and biomarkers.
Read more
At the moment, the platform offers standardin vivo experimentation procedures for the evaluation of new therapeutic targets, nanotherapies and biomarkers in the field of oncology and rare diseases.
Read more
Latest
news
News
Dr. Simó Schwartz Jr. assumes the general direction of the Blood and Tissue Bank after 16 years at VHIR
Dr. Simó Schwartz Jr. has been appointed as the new general director of the Blood and Tissue Bank, replacing Dr. Enric Argelagués, who leaves his position today...
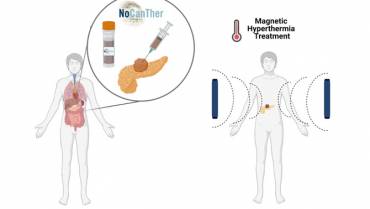
Read moreVall d’Hebron leads a clinical study testing the use of nanoparticles for the treatment of locally advanced pancreatic cancer
Nanomedicine has emerged as a potential therapy for pancreatic cancer, a disease with a 5-year survival rate as low as 5%. The international project NoCanTher was...
Read moreNanomedicine & Bioengineering
Publications
Thermo-Responsive Hydrogels for Cancer Local Therapy: Challenges and State-of-Art.
Rafael D, Merce Roca Melendres, Andrade F, Montero S, Martinez-Trucharte F, Vilar-Hernandez M, Francisco Duran-Lara, Schwartz S Jr, Abasolo I Int J Pharm. 2021 Jul...
Read more