Renal Physiopathology
A major focus of our laboratory has been to investigate the role of androgens in kidney pathophysiology, by identifying androgen-regulated genes whose expression is restricted to the proximal tubule cells of the kidney.
The molecular mechanisms that control specific expression of those genes in tubular epithelia have been studied in different mouse models and in androgen-responsive proximal tubule derived cell lines. Some of these novel identified genes have also been investigated at the functional level. The interaction found between the kidney androgen-regulated (KAP) gene and Cyclophilin B (CypB), one of the receptors of the potent immunossupressant Cyclosporine A (CsA), prompted us to investigate the molecular and cellular mechanisms underlying kidney tubular injury induced by renal nephrotoxicants and isquemia-reperfusion processes. The role of KAP, CypB and other members of the immunophylin family in processess related with kidney injury and regeneration are currently being investigated by using genomic approaches in proximal tubule derived cell lines and in Tg and KO mice. Recent data from our laboratory has shown that Tg mice overexpressing the KAP protein in proximal tubule cells develop hypertension mediated by oxidative stress and focal segmental glomeruloesclerosis. We are currently working with this Tg model and producing KAP KO mice to further investigate the role of KAP in renal pathophysiology. Another gene of interest is the one coding for the hepatitis A viral receptor (hHAVR), first identified in our laboratory by its ovreexpression in clear cell renal cell carcinomas (ccRCC), the most malignant and frequent form of renal cancer that arises in proximal tubule cells and is more prevalent in men than women. We are currently investigating the role of hHAVR in the development and progression of human ccRCC.
An important part of the group’s efforts is focused to the identification of early, specific and sensitive biomarkers of renal dysfunction by menas of high-throughput proteomic analyses in urine and blood samples of trasplanted patients under different immunossupresant regimes. These techniques are also used for the identification of putative plasma permeabilizating factors in patients suffering idiopatic non-familial focal segmental glomeruloesclerosis and in ccRCC patients.
Our close relationship with nephrologists, urologists and pathologists from our Institution promotes collaborations aiming towards the identification of potential biomarkers and therapeutical targets that migth be useful for future clinical interventions. Finally, the possibility of using nanoconjugates for drug delivery opens new perspectives for targeted therapy.
These are currently the main lines of research at the kidney phisipathology Group:
- Role of Hepatitis A viral receptor (HAVR) / kidney injury molecule-1 (KIM-1) in the development and progression of clear-cell renal carcinoma (ccRCC), as well as, in the renal tubule injury/regeneration processes.
- Overexpression of this protein in 60% of the ccRCCs has already been described. HAVR/KIM-1 overexpression in human ccRCC cell lines blocks cell differentiation and promotes cell scattering. We aim to determine the role of HAVR/KIM-1 in the development and progression of ccRCC, and its possible value as a diagnostic and prognostic biomarker.
We also focus on KIM-1’s role in ischemia/reperfusion- or nephrotoxic-induced renal tubular injury. Overexpression of this protein in kidney injury has been described. However, whether its involvement is associated with processes enabling to recover tubular epithelium or potentially increasing damage is not known to this date. With the assistance of cultured renal tubular cell models, we are now investigating whether KIM-1 expression shifts are correlated with renal proximal tubule regeneration ability and, as a consequence, investigating its potential therapeutic application.
Androgen activity in renal pathophysiology. Identification of androgen-regulated kidney-specific genes and their role in the pathogenesis of renal, cardiovascular disease and metabolic disorders.
Among the genes identified in our laboratory that are kidney-specific and regulated by androgens at the transcriptional level we are particularly focused on the one that codes for the kidney androgen-regulated protein (KAP). Besides characterization of the functional promoter elements that enable KAP expression in proximal tubule epithelial cells, we have generated a transgenic (Tg) mouse model that overexpresses KAP in proximal tubule cells under the presence of androgens, in order to mimick the endogenous KAP expression pattern. KAP Tg mice show altered lipid metabolism, glycosuria, proteinuria and hypertension, as well as focal segmental glomerulosclerosis mediated by increased oxidative stress. We are currently working in this Tg model and also preparing conditional knock-out mice to further caharacterize the role of KAP in renal pathophysiology.
Pathologic mechanisms leading to chronic allograft disease and its potential mediators. Detection of early markers of the chronic kidney disease of the graft.
Chronic allograft nephropathy (CAN) is one of the major causes of graft loss in kidney-transplanted patients. The pathogenetic mechanisms of CAN are probably multifactorial, including early noxious agents as a consequence of ischemia / reperfusion of the graft or high loading doses of anticalcineurinics (aCN), and also chronic damage following aCN therapy, rejection or other reasons. We want to determine the proteomic and genomic changes occurring in tubular cells after different noxious agents (cyclosporin, tacrolimus, other renal toxicants, hypoxia), and also the effects caused by immunophilin silencing (anticalcineurin receptors) in the renal proximal tubule cells. Our objective is to identify specific markers of kidney injury that would be useful to anticipate toxicity or injury in early starges. Those putative markers will be clinically validated in collaboration with the Nephrology and the Pathology services of Vall d’Hebron Hospital.
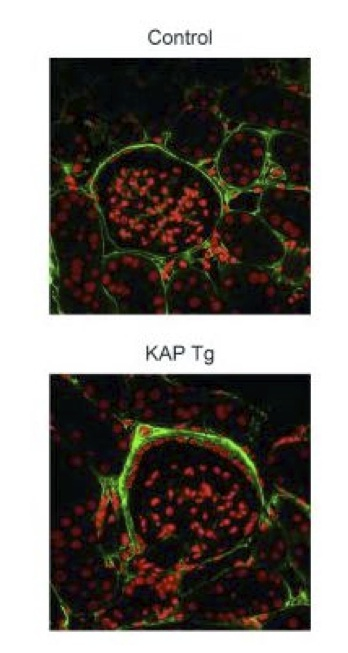

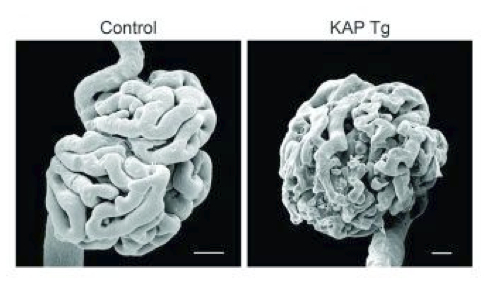
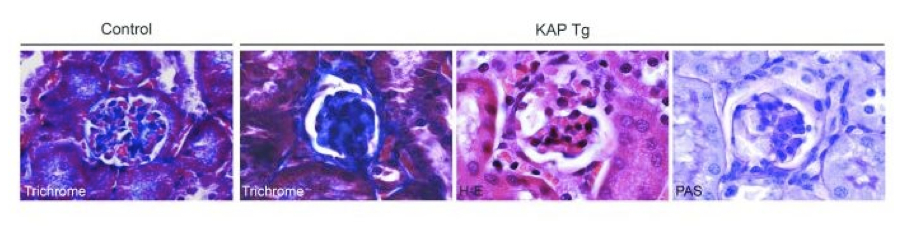
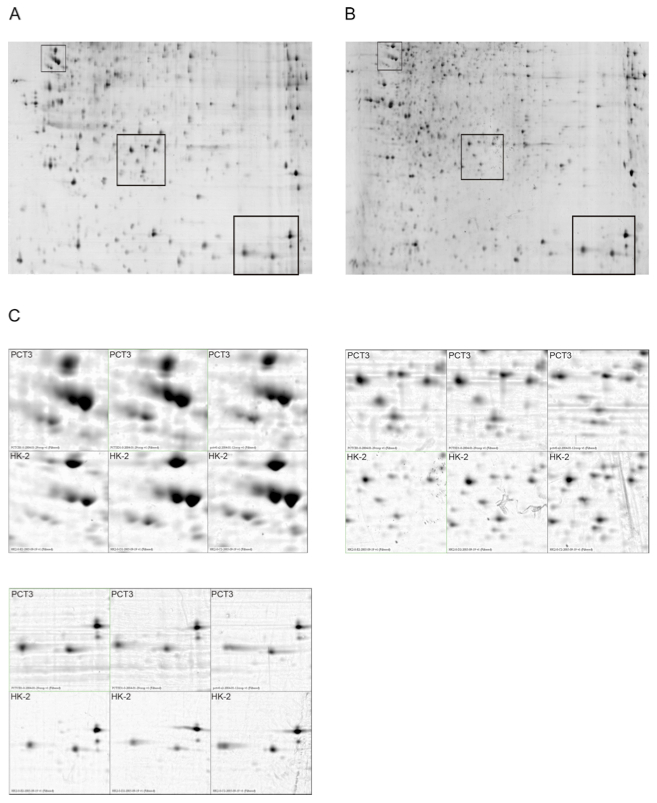
Focal segmental glomerulosclerosis
Idiopathic nonfamilial focal segmental glomerulosclerosis (FSG) is a disease with no treatment, whose usual outcome is end-stage renal disease frequently recidivating after transplantation. In close cooperation with the Nephrology and Paediatric Nephrology services of Vall d'Hebron hospital together with hospitals throughout the country that provide a significant number of patients, we intend to identify the hypothetical blood factor that causes the proteinuria observed in this disease. Identification of such plasma factor, by means of differential proteomic analysis, would allow the definition of therapeutic targets for the disease, which currently lacks an effective treatment. Our second objective is to find biomarkers that enable us to foresee a potential recidivation and the consequent loss of the graft following renal transplantation to FSG patients.





