Drug Delivery and Targetting
Dr Simó Schwartz Jr (1967th, Barcelona) obtained his Medical degree in 1991 at the Faculty of Medicine of the Autonomous University of Barcelona, where he also got his PhD in 1996 working on oncogenic signalling pathways in prostate cancer. He was a research fellow in the New York Univ. Medical Center in 1993, working on molecular mechanisms related to Ras/Raf activation in transgenic mice. In 1996 he moved to California as postdoctoral research at the Burnham Institute for Biomedical Research, where he work intensively under the guidance of Dr Manuel Perucho in the molecular pathways involved in the development of colorectal cancer. In 2000 he was appointed as head of the Molecular Oncology and Aging laboratory at the Molecular Biology and Biochemistry Research Center (CIBBIM) of the Vall d’Hebron University Hospital in Barcelona. He is nowadays a Board member of the CIBBIM and also member of the Science Committee of the Vall d’Hebron Research Institute.
In 2004, Dr Schwartz start few collaborations with biotech companies in the field of diagnostic and prognostic biomarkers and new therapeutic targets in colorectal cancer. The success of these collaborations lead to 6 patents transfered in the last four years to leading companies of the biotech and pharma sectors. Also, the development of several research projects among which are worth to mention projects of the National R+D Grants, CENIT (industrial consortiums) and few european and international innitiatives. Since 2006 he is a member of the Science Advisory Board of Oryzon Genomics, a Spanish leading biotech company. Recently (2007), he has been appointed as Coordinator of Nanomedicine at the CIBBIM (now CIBBIM-Nanomedicine) and coordinator of its strategic action plan for R+D, which is focused on the research of new biomedical nanotechnology-based applications. In particular, new drug delivery systems, image based diagnostic systems and preclinical validation of therapeutic conjugates and bio-nanosensors, mainly in the areas of oncology and rare diseases.
He also leads the “drug delivery and targeting group” at the CIBBIM-Nanomedicine. In this context, Dr Schwartz Jr is coordinator and collaborator of several research projects directly related with the obtention of therapeutic drug delivery systems. Among them are the CENIT “Oncnosis” project, two ERANET projects (IMMAPROT on Industrial biotechnology – ERA-IB; NANOSTEM – EuroNanoMed, coordinated by him), and an international project of the Iberian Nanotechnology Institute (OncoNanoTarget). All of them projects involving SME’s in which animal models are being used for preclinical validation of new therapies directed against tumor cells. Dr Schwartz Jr is also member of the Nanomedicine Spanish Platform (NanomedSpain) and of the “European Platform for Nanomedicine” where he co-authors the 2006 Research Strategic Agenda intended to the European Commission. His research group is also a group member of the “CIBER de Bioingeniería, Biomateriales y Nanomedicina” (CIBER-BBN) of the Spanish Health Institute CarlosIII (ISCIII) which gathers a total of 50 research groups of national excellence in the field of nanotechnology and nanomedicine. Since 2007, Dr Schwartz Jr acts as the Nanomedicine Coordinador of CIBER-BBN at the national level where he also leads two intramural projects and collaborates in other seven coordinated projects. All of them are centered in biomedical applications of nanotechnology and nanomedicine for human health.
Objectives:
The group on Drug Delivery and Targeting seeks two main goals; on the one hand, the identification of new disease biomarkers and therapeutic targets, with special focus on cancer molecular pathways; and on the other hand, the development of new delivery strategies in applied nanomedicine, with a particular interest into new delivery and targeting approaches for clinical applications.

Research lines:
1. Identification of new disease biomarkers and therapeutic targets
There are several research lines dedicated to the study of oncogenic molecular pathways related with i) genomic and microsatellite instability in gastrointestinal tumors; ii) condensin complexes in colorectal tumorigenesis, iii) molecular alterations caused by defects in the DNA repair system (basically, mismatch repair –MMR- pathway and repair of double strand breaks –DSB-) and their relation with tumor resistance to chemotherapy, and iv) identification and validation of new biomarkers and therapeutic targets by means of high-throughput screening (HTS).
i) Genomic and microsatellite instability in gastrointestinal tumors
This project aims to explain the striking contrast in survival and metastatic capacity between chromosomal instable (CIN, 85% incidence), and microsatellite instable (MSI, 15% incidence) colorectal carcinomas, of the different aggressiveness among tumors bearing diverse K-ras point mutants, and of the distinct mutational selectivity of the K-ras and B-raf oncogenes. To that purpose, whole-body optical imaging will be used to perform a longitudinal analysis of metastatic dissemination, as well as an evaluation of the requirement for K-ras and/or B-raf oncogene expression in maintaining metastatic foci growth. This model will allow the dissection of the molecular pathways activated by B-raf and K-ras mutants, as well as the transition from dormant micrometastases to expansive metastases at the different target sites.
ii) Condensin complexes in colorectal tumorigenesis.
We focus into the involvement of chromatin remodelling and chromosomal condensation complexes and protein partners in the development of colorectal tumors and cancer progression. We also address functional studies related to the involvement of these complexes in gene transcriptional regulation and their interactions with DNA repair complexes and the histone code.
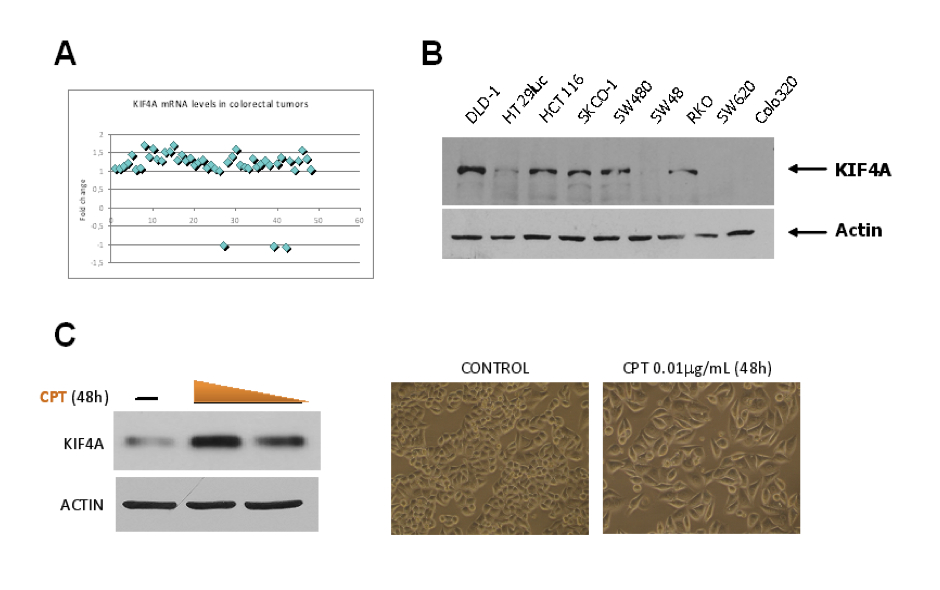
Figure 1: Condensin interactors. Expression and its sensitivity to camptothecin. A) Condensin-interactor was found to be overexpressed in 24 out of 48 colorectal cancer (CRC) samples analyzed by expression microarrays. B) protein expression was further confirmed by Western Blot analysis in a panel of CRC cell lines. C) Furthermore, Camptothecin (CPT), a drug commonly used in the treatment of CRC, increased the expression of the protein in HeLa cells, suggesting that this interactor could be used as aresponse prediction factor for CPT treatments
ii) DNA repair system and tumor resistance to chemotherapy
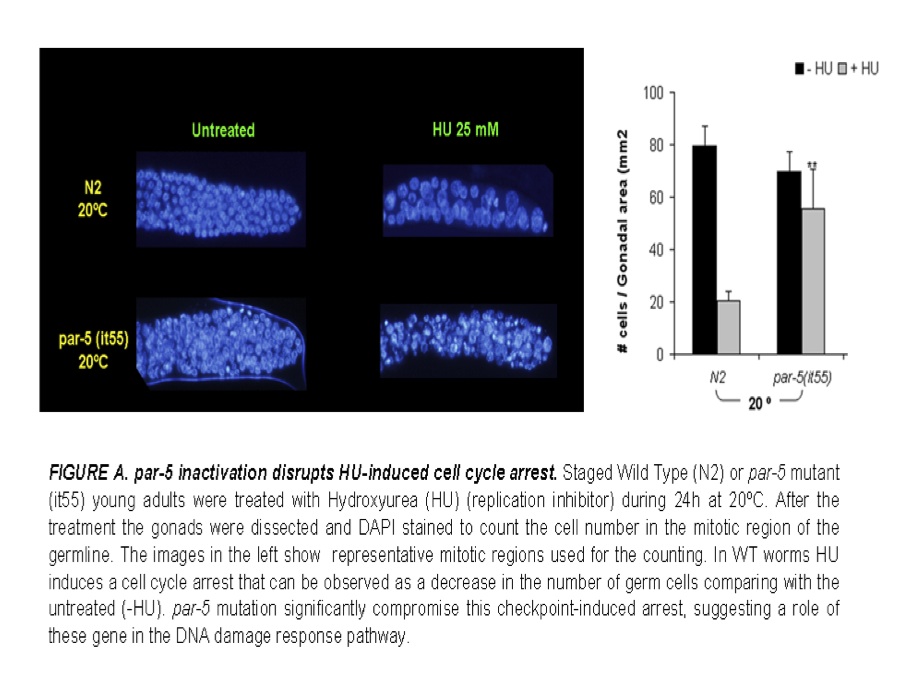
This research line is focused into the identification of new signal transducers involved in DNA damage control pathways which are the main responsible of controlling cell death and repair mechanisms. We are also studying their alterations and biological involvement in tumor cell development and metastasis spread, together with the consequences excerted at the level of tumor treatment response to classical and non-classical chemotherapy. Validation of new checkpoint targets are also done in C.elegans models by using hydroxyurea treatments and specific transgenic mutants and siRNA of target genes.
2. Applied Nanomedicine: new drug delivery and targeting strategies for biomedical applications
We focus into new targeting strategies to ensure a specific delivery of therapeutic compounds into the most appropriate target cell to improve treatment response and achieve lower toxicity and higher therapeutic activities in several human diseases, with particular interest in delivery of chemotherapeutic drugs to cancer cells and enzyme replacement therapies for rare diseases. In addition, alternative targeting strategies are being designed to improve imaging-based diagnostics by using new cell-targeted nanoconjugates.
Some of the projects explained bellow are developed in collaboration with other groups at CIBER-BBN (Centro de Investigaciones en Red en Biomateriales, Bioingenería y Nanomedicina, Instituto de Salud Carlos III) or within specific European consortiums.
i) Enzyme replacement therapy for storage diseases: new therapeutic strategies
Fabry disease is an X-linked recessive disorder caused by a deficiency of lysosomal hydrolase α-galactosidase A (GLA). This enzymatic defect causes the progressive cellular accumulation of neutral glycosphingolipids, giving rise to a multisystemic clinical symptomatology. Current enzyme replacement therapy (ERT) has a limited treatment efficacy in patients with advanced stages of the disease. The objective of this research line is to improve the ERT by using new therapeutic compounds (nanoparticles or specifically designed proteins) of GLA targeted to the endothelial cells, one of the main cell type affected by GLA substrate accumulation. In addition, we also collaborate in a project focused on the validation of new integrated, multi-host approaches for the improved microbial production of high quality GLA enzymes for industrial purposes (IMAPPROT).
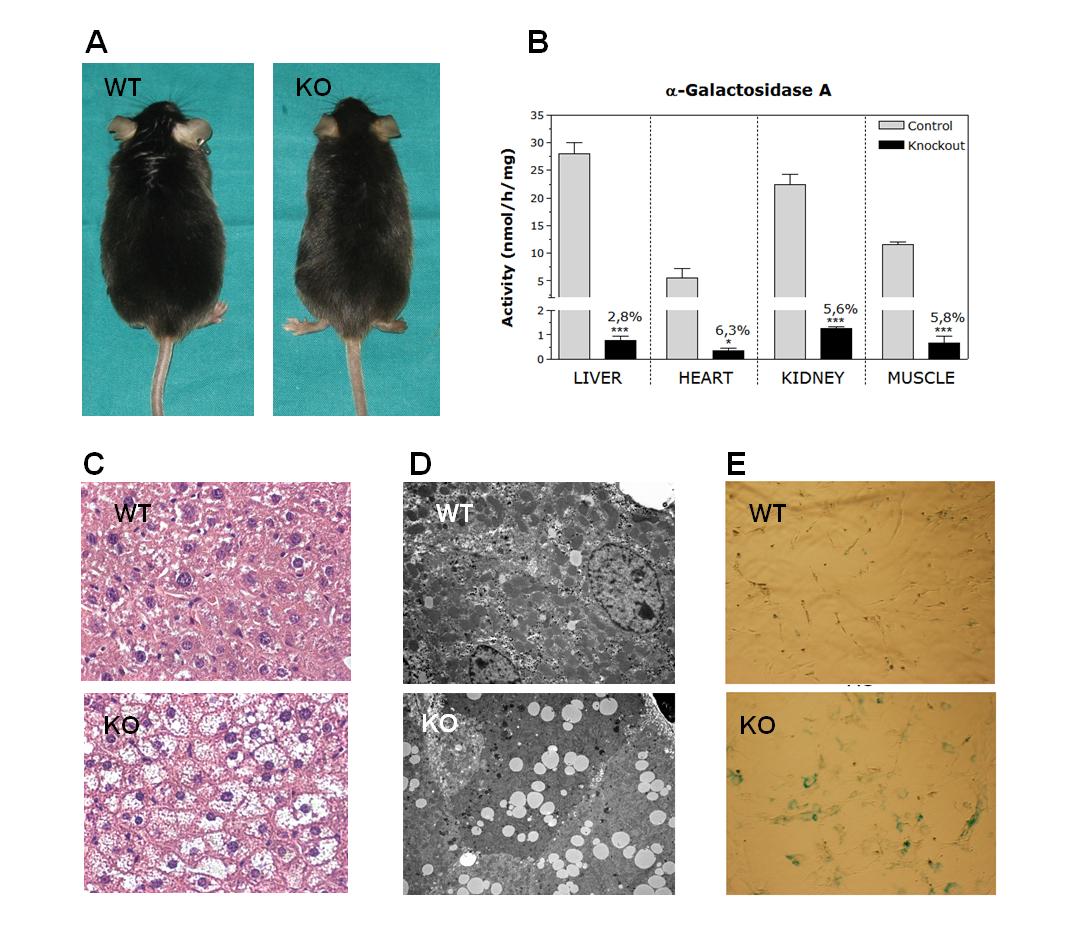
Figure 2: Characterization of the Fabry KO mice. A) Overall phenotype, B) GLA activity, C) Liver histology, D) Lysosomal deposits shown by electronic microscopy, and E) Senescence (b-Gal staining) in primary endothelial cultures.
ii) NANOSTEM: Targeting Combined Therapy to Cancer Stem Cells
In many solid tumors, resistance to therapy and metastatic disease seem to be sustained by the presence within the tumors of the cancer stem cells (CSC) capable of regenerating a tumor after chemotherapy and/or radiation treatment. In breast cancer, these cells correspond to a small fraction of cells within the tumor that express stem cell markers (CD44+/CD24-/low/lin-) which provides a useful target to the delivery of therapeutic agents to CSC. In this network project some of the partners will focus into the design of specific vehicles for the simultaneous deliver of chemotherapeutic drugs and/or shRNAs with known antitumor activity to breast CSC. To this end, different types of nanoparticles will be directed to the CSC compartment by using the CD44 receptor as a target. Such systems will allow specific CSC-targeting, and together with the enhanced retention an permeability effect (EPR) will improve accumulation of drugs into the tumor area and should yield better therapeutic response. At CIBBIM-Nanomedicine, therapeutic activity, nanoparticle internalization and toxicology of these nanoparticulated systems will be addressed by using adequate in vitro and in vivo CSC models.
iii) ONCONANOTARGET: Advancing the field of drug delivery - combined targeted treatments against human breast cancer and human leukemia
The idea in the ONCONANOTARGET Network is to selectively abrogate tumour-protective functions aiming at either improving sensitivity of tumour cells to chemotherapy or finding synergistic combinations that may improve the clinical outcome for the treatment of breast cancer or leukaemia patients.
Therefore, the main objectives of this project are: (i) To design and characterise ligand-targeted nanosystems for nucleic acid (siRNA) and drug delivery; (ii) To compare, in vitro, the gene-silencing efficiency of the developed targeted lipid-based or polymeric-based nanosized systems containing nucleic acids against Bcl-2 oncogene in breast cancer leukaemia and cells; (iii) To evaluate the cytotoxic activity of individual treatments with either gene silencing with targeted system previously selected or targeted polymer-anticancer drug conjugate as compared to combined treatments (targeted gene silencing combined with targeted polymer-anticancer drug conjugates) against leukaemia and breast tumour cells; and (iv) Therapeutic evaluation of the treatment modality previously selected in an animal model of human breast cancer. In this project, in vivo proof of concept will be limited to breast cancer.
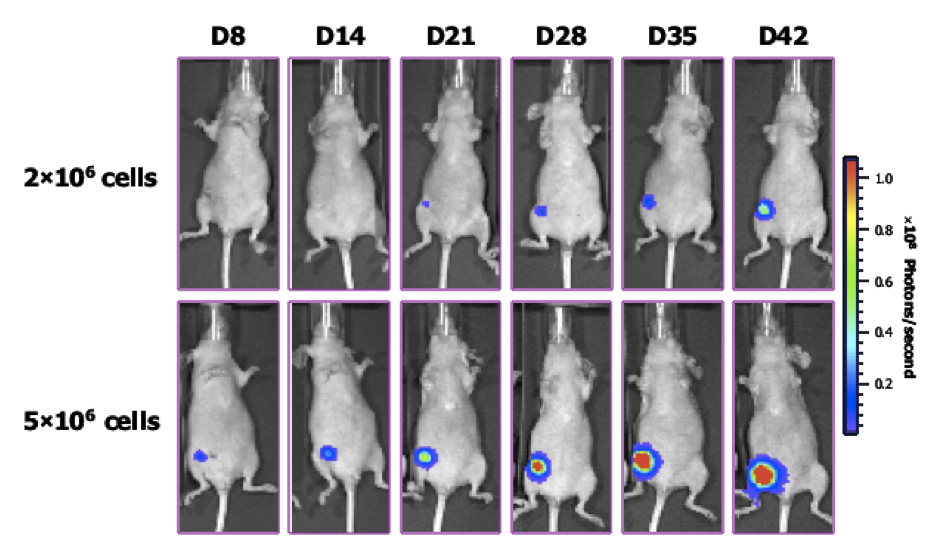
Figure 3. Orthotopic mammary fat pad tumor growth and spontaneous metastases of breast cancer cells with luciferase overexpression. Female mice received a orthotopic injection of 2 or 5x106 MDA-MB-231 cells and bioluminescence tumor growth was monitored over time using the IVIS® Spectrum This same model will be used for the therapeutic evaluation of the nanosystems developed within the ONCONANOTARGET Network.
iv) Treatment of advanced colorectal cancer by novel drug delivery systems, sensitive to metalloproteinases
Current chemotherapeutic treatment for colorectal cancer implies the use of high doses of cytotoxic medicaments, specifically adjuvant combinations of 5-fluorouracil and Irinotecan, which bring many adverse effects to the affected patient. This project proposes a program centred on the development of new nanomedicines, based on polymers of multifunctional character that brought together different chemotherapeutic agents, allowing a combined double or triple therapy using much lower systemic doses and significantly reducing undesirable side-effects. In this case, we will focus on increasing these advantages with the utilization of synthetic peptides sensitive to degradation by matrix metalloproteases (MMP), which will bind the polymeric nanocarrier to the chemotherapeutic drug. The activity of MMPs will favour the liberation of the drug and its activity in MMP rich environments, so to say primary tumors and metastatic sites. The project includes the processes of synthesis, chemical characterization and optimization of the nanomedicines, as well as their in vitro and in vivo validation.





