DIAGNOSTIC NANOTOOLS for Fast Assay and Biosensor Development (DINA)
Objectives:
One of the greatest challenges in global healthcare is the lack of adequate diagnostic tools for early disease detection and adequate treatment monitoring. Accordingly, the development of low-cost, rapid, sensitive and robust assays and sensors has been a priority for the healthcare sector and the research community. It is for this reason that the recent advances in rapidly evolving fields, such as Nanotechnology and Biosensor development, have raised great expectations and bitter controversies. DINA is brought to live in 2014 with a main goal: the exploitation of nanostructures, nanomaterials and nanocomponents as transducers, biofunctionalization platforms and signal amplifiers for fast assay and biosensor development. But always with a clear objective: the production and/or improvement of realistic diagnostic tools that could be effectively used at a diagnostics laboratory.
Electrochemical Biosensors as diagnostic tools.
By definition, a biosensor is a bioanalytical device that incorporates a molecular recognition element (bioreceptor) associated to, or integrated with, a physicochemical transducer. According to this, a biosensor is formed by three components: a bioreceptor able to capture specifically the target of interest, a signal transducer able to convert target binding into a measurable electrical signal, and a signal processor that quantifies, analyzes and displays the results. In this way, analyte biocapture can be directly translated into a measurable signal. To date, numerous biosensors have been developed using a wide variety of biorecognition elements (ranging from “classical” bioreceptors such as antibodies, nucleic acid probes, antigens or enzymes, to novel alternatives like aptamers, biomimetic polymers or phage displayed peptides) and types of transducers (principally clustered into optical, electrochemical and micromechanical).
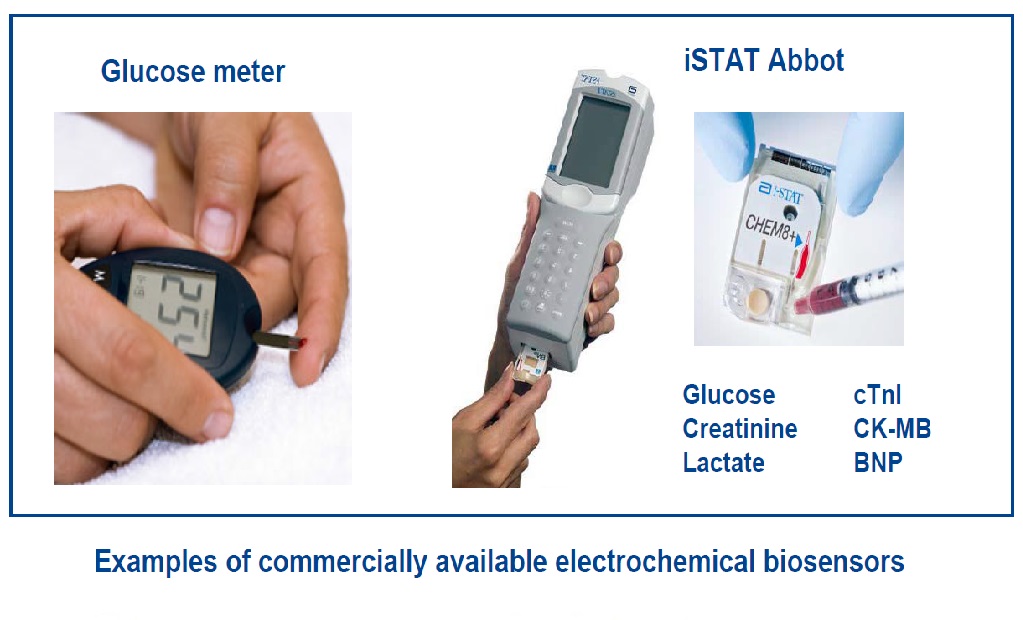
Electrochemical biosensors are among the most commonly used nowadays thanks to their portability, cost effectiveness, small size, rapidity, and robustness. Additionally, electrochemical transducers and equipment are relatively easy to miniaturize into multiplexed platforms, which coupled to integrated microfluidics is highly compatible with multi-analyte testing. This favours the development of point-of-care (POC) devices, to be used directly by the patient or at the surgery. The two clearest examples are probably the glucose electrochemical biosensor, which has facilitated life to diabetic patients to a great extend, and the hand held i.STAT clinical analyser (Abbot), which combines several electrochemical biosensors on a single chip and is used for multiple electrolytes and metabolites in clinical samples.
Nanocomponents and Nanostructured Materials in Fast Assay and Biosensor Development.
Nanotechnology is a rapidly emerging field that is having an enormous impact on assay and biosensor development and, by extension, in their application to diagnostics. Nanomaterials can display almost unlimited combinations of composition, size, dimensions and shape, which can be tailored and coupled to bioreceptors in order to produce nanoprobes with desired properties. Since nanostructures are characterized by high surface to volume ratios, they have been extensively used as multi-label carriers for signal amplification. In opposition to detection using bioreceptors conjugated to a single label unit, multi-label nanoparticles have been claimed to provide higher and faster responses, contributing to improved bioassay/biosensor detection limits by up to three orders of magnitude.
Alternatively, a variety of nanomaterials, such as fullerene derivatives, gold nanoparticles, rare earth nanoparticles and ferromagnetic nanoparticles, have been explored as artificial enzymes or enzyme mimics (nanozymes) thanks to their intrinsic non-enzymatic catalytic activity for a variety of molecular substrates. Used as labels in bioassay/biosensor development, nanozymes appear to be more stable and cheaper alternatives than natural enzymes. A particular example is nanoparticle-induced metal deposition. For instance gold nanoparticles can selectively reduce silver and gold salts to silver or gold, respectively, in the presence of a reducing agent. This results in significantly increased size which affects directly electrode transduction.

Since their discovery, carbon nanotubes (CNT) have been exploited for a number of electroanalytical and sensing purposes. Among others, CNT incorporation onto an electrode allows taking advantage of the high mechanical resistance, chemical stability and electronic conductivity of this nanocomponent. For instance, CNT-modified electrodes exhibit active surfaces of increased roughness and area, electrocatalytic activity towards a wide variety of molecules, and faster electron transfer than unmodified electrodes. The simplest strategy for the production of CNT-modified electrodes is the deposition of a small volume of CNT dispersion onto the electrode surface followed by solvent evaporation, but a variety of alternative strategies have been reported.
We recently demonstrated that CNT dispersed in aqueous media have a strong tendency to adsorb non-specifically onto the surface of magnetic particles (MP). The resulting MP/CNT composite can then be arranged onto an electrode using a magnet, which is extremely fast, simple and reversible, allowing easy electrode regeneration/reutilisation. CNT magnetic co-entrapment can then serve to produce nanostructured electrodes of enhanced performance, but has been also applied to the electrochemical monitoring of the MP surface itself. In this context, CNT serve as wires that connect MP between them and with the electrode and allow the electrochemical detection of any electroactive targets and labels that have been previously bound to the MP surface. For instance, we have shown that dopamine, an electroactive neurotransmitter, could be first immunocaptured and concentrated using specific MP, which eliminates interference by non-targeted sample components, followed by CNT magnetic co-entrapment onto an electrode, which promotes surface electrical wiring and allows straightforward electrochemical sensing of the captured molecule.





